The forbidden energy gap in germanium is 0.7 eV. The wavelength whose absorption is done by germanium is:

The forbidden energy gap in germanium is 0.7 eV. The wavelength whose absorption is done by germanium is:

Electron removed1 ^st electron2 ^nd electron3 ^rd electron4 ^th electron5 ^th electron6 ^th electronIonization energy(kJ/mole) 733 1,450 7,730 10,538 13,618 18,101 The ionization energies (potentials) for the removal of different electrons from

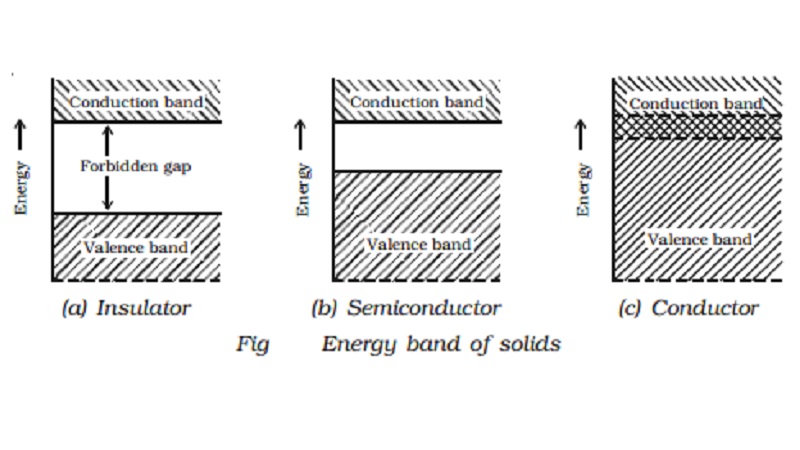
The energy gaps (E) between the valence band and conduction band, g for diamond, silicon and germanium are in the order:

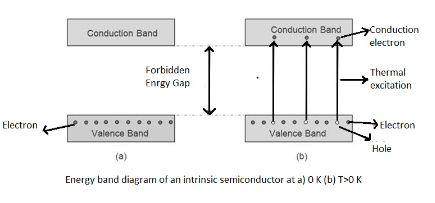
The forbidden energy gap of Ge is 0.75 eV, the maximum wavelength of incident radiation - producing an electron - hole pair in germanium semiconductor is:

Forbidden energy gap of Ge is `0.75eV`, maximum wave length of incident radiation for producing elec - YouTube
In germanium crystal, the forbidden energy gap in joule is:A. $1.6 \\times {10^{ - 19}}$B. ZeroC. $1.12 \\times {10^{ - 19}}$D. $1.76 \\times {10^{ - 19}}$

The forbidden energy gap of Ge is 0.75 eV, the maximum wavelength of incident radiation - producing an electron - hole pair in germanium semiconductor is:
![PDF] Experimental estimation of the band gap in silicon and germanium from the temperature–voltage curve of diode thermometers | Semantic Scholar PDF] Experimental estimation of the band gap in silicon and germanium from the temperature–voltage curve of diode thermometers | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2a991f16c0139e0c55c0f366537e194ea5319683/2-Figure2-1.png)
PDF] Experimental estimation of the band gap in silicon and germanium from the temperature–voltage curve of diode thermometers | Semantic Scholar
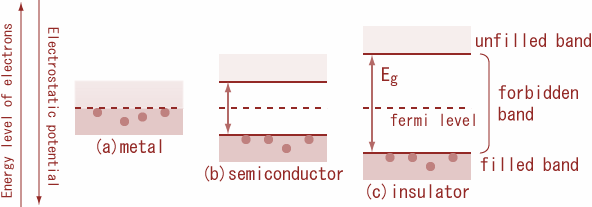
Carbon, silicon and germanium have four valence electrons each. These are characterised by valence and conduction bands separated by energy band gap respectively equal to ${\\left( {{E_g}} \\right)_C},{\\text{ }}{\\left( {{E_g}} \\right)_{Si}}{\\text ...

Electron removed1 ^st electron2 ^nd electron3 ^rd electron4 ^th electron5 ^th electron6 ^th electronIonization energy(kJ/mole) 733 1,450 7,730 10,538 13,618 18,101 The ionization energies (potentials) for the removal of different electrons from

The forbidden energy gap of germanium is 0.72eV. What do you understand by it? | 12 | ELECTRONIC... - YouTube